EFFICACY IN THE TREATMENT OF DEPRESSIVE DISORDERS

Taysa Leite de Aquino1, Jaiany Rodrigues Libório1, Ana Luiza Menezes Santana Bezerra1, Giovana Macêdo Egídio Cavalcante1, Dara Fernanda Brito Duarte1, Ana Luíza de Aguiar Rocha Martin1, Juliane dos Anjos de Paula1, Modesto Leite Rolim Neto1
1 School of Medicine of Juazeiro do Norte – FMJ/Estácio, Juazeiro do Norte, Ceará, Brazil.
Corresponding Author
Modesto leite Rolim Neto
School of Medicine of Juazeiro do Norte – FMJ/Estácio, Juazeiro do Norte, Ceará, Brazil
Phone: (+55 88 99042979)
Email: modestorolim@yahoo.com.br
ABSTRACT
Introduction: Depression, being the most disabling disease in the world and presenting high morbidity and mortality, requires an effective treatment to ensure the maintenance of the patients' quality of life. About a third of patients do not respond adequately to antidepressant treatment. New pharmacological therapies are being approached in order to reduce the unfavorable outcomes resulting from this pathology.
Objective: To develop a systematic review of the literature on the new possibilities of pharmacological treatments for depression in the last four years.
Method: A systematic review was carried out following the PRISMA protocol (Preferred Reporting Items for Systematic Reviews and Meta-Analysis). We have included studies on the effectiveness of new pharmacological therapies in patients with major depressive disorder and treatment-resistant depression.
Results: In the treatment of the major depressive disorder (MDD), the use of brexpiprazole 1 mg/day in addition to the usual antidepressant treatment (ADT) resulted in significant improvement in the main symptoms. Already referring to treatments for treatment-resistant depression (TRD): nasal esketamine proved beneficial in the short term, intravenous (IV) ketamine also appears as an effective therapy.
Conclusion: The use of brexpiprazole 1 mg/day associated with an antidepressant, and the use of lactobacilli represent a new option for those with MDD. In addition, both ketamine, riluzole, and cariprazine appear as new hopes for those who have not yet achieved remission of the depressive disorder with the use of more than two antidepressants.
Key-words: New Treatment; Pharmacological; Depressive Disorder.
1.INTRODUCTION
The World Health Organization (WHO) already predicted, according to the pace of diagnosis and the characteristics of the disease, that in 2020 depression would become the most disabling disease in the world. Currently, depression is responsible for the reduction of up to ten years of life expectancy, is the main cause of disability, has high rates of morbidity and mortality, being responsible, therefore, for an extremely negative direct impact on the quality of life of more than 322 million of people around the world, confirming and exceeding WHO estimates. (Hudgens et. al., 2021; Daly et. al., 2019; World Health Organization 2017).
Facing a disease with unfavorable outcomes, treatment becomes essential to maintain the patients' quality of life. However, a third of individuals diagnosed with depression do not respond adequately to the first-choice treatments, including monoamine reuptake inhibitors (Zhuo et. al., 2019). In addition, these drugs take between three to seven weeks to start their antidepressant effects and up to 12 weeks to achieve maximum effect, as well having side effects in most users. In many situations, the combination of two or more of these drugs is not able to cease the patient's depressive symptoms, raising him to the diagnosis of treatment-resistant depression (Griffiths et. al., 2021; Hudgens et. al., 2021). In this sense, the need for more effective treatments, with a faster onset of action, better dosage, and fewer side effects has increased in recent years and new therapeutic approaches for depressed patients have emerged aiming to reduce the unfavorable outcomes resulting from this pathology.
However, the proposed therapies face difficulties due to the long time spent and the high costs for the development of a new drug. This path takes, on average, 14 years. However, with the study of new approaches to products that are already consolidated in the market and the reuse of drugs, this time reduces to less than half, around six years (Fabbri et. al., 2020). Currently, many drugs are in the process of analysis and face difficulties such as the short observation time, the need for further studies related to the neurobiological mechanisms involved in the pharmacodynamics of the proposed drugs; or even the difficulties with the selection of the requirements for the study, which is often small and restricted to a region. However, both a long-term analysis and an understanding of the mechanism of action of the drug associated with the physiological processes of depression and the heterogeneity of the study group are extremely useful to understand and attest to the effectiveness of new therapies (Zhuo et. al., 2019; Griffiths et. al., 2021).
Given this situation and taking into account that 20 to 30% of individuals with major depressive disorder (MDD) develop treatment-resistant depression (TRD) (Fabbri et. al., 2020), we aim to develop a systematic review of the literature regarding the new possibilities of pharmacological treatments for depression based on the acronym PICO: P: study population - people with depressive disorder according to DSM V 296.21, 296.22, 296.23 and 296.24; I: intervention – to demonstrate the effectiveness of new pharmacological therapies for depression; C: control - individuals with MDD and TRD; O: outcome - new pharmacological approaches to depressive disorders.
This question has had a greater impact in recent years due to the need for new pharmacological treatments since the number of patients with TRD and MDD who respond inadequately to conventional treatment is increasing. Thus, this study aims to formulate a systematic review of the literature regarding new pharmacological treatments for depression.
2 METHOD
A systematic review was carried out, following the PRISMA protocol (Preferred Reporting Items for Systematic Reviews and Meta-Analysis).
2.1. Inclusion criteria
The inclusion criteria were studies published in English, Portuguese and Spanish, suitability for this review, methodological rigor applied, and full text available for free. Review articles, as well as comments on literature, editorials, communications, and letters to the editor, were excluded. We had no restrictions related to the study design, methodological quality, or language. The problem with the language was solved through a reasonable degree of comparability, which allowed us to systematically analyze the selected evidence, its critical evaluation process, and its success in including relevant studies in other languages.
We classified assessments according to their level of inclusion of studies in other languages. Reviews that excluded non-English studies with an explicit justification in the research question or research objectives were categorized as justified by R1 (that is, justified in English), while those that excluded non-English studies without justification were categorized as restricted to RR1 (that is, languages that are not restricted to English). Reviews that did not explicitly exclude studies that were not in English were categorized as RR1-open unless they successively included studies that were not in English, in which case they were RR1-inclusive. Finally, revisions that did not declare language criteria were considered to be RR1-open.
2.2. Literature search and article selection
To search for the studies, the following databases were used: PubMed, LILACS and Science Direct data, from 2018 to 2021c with the descriptors MeSH and DeSC: “Pharmacological Treatment” AND/OR “Tratamento Farmacológico”, “New Treatments” AND/OR “Novos Tratamentos”, “Depressive Disorder” AND/OR “Transtorno Depressivo”.
2.3. Data extraction and methodological quality assessment
One researcher (MLRN) extracted the data, and another verified the extraction. Two researchers (ALARM and JAP) independently assessed the methodological quality of systematic reviews using the AMSTAR tool and qualitative studies using the CASP checklist (Critical Appraisal Skills Program - CASP, 2020). One researcher (ALARM) assessed the quality of cross-sectional studies using the JBI Prevalence or the JBI Cross-sectional analytical checklist and longitudinal studies using the JBI Cohort checklist (Johanna Briggs Institute).
2.4. Data presentation and analysis
We describe the interventions and results based on the information provided in the studies. When studies showed results of new pharmacological therapeutic approaches to depressive disorders in numbers without numbers, we extracted them using online software (https://apps.automeris.io/wpd/).
2.5. Data presentation and analysis
We summarize the results narratively. We describe the interventions and results based on the information provided in the studies. When the studies showed results regarding new pharmacological therapeutic approaches for depressive disorders in the last four years, we extracted them using online software (https://apps.automeris.io/wpd/). We classify the certainty of the evidence using the GRADE approach (Grading of Recommendations, Assessment, Development and Evaluations).
3. RESULTS
The data were collected independently by three reviewers (ALARM, JAP, ALMSB, and MLRN). If there was disagreement between the evaluators on the inclusion and exclusion criteria, a specific discussion was made about the article in question until a final consensus. The necessary information was collected from published articles (Figure 1)
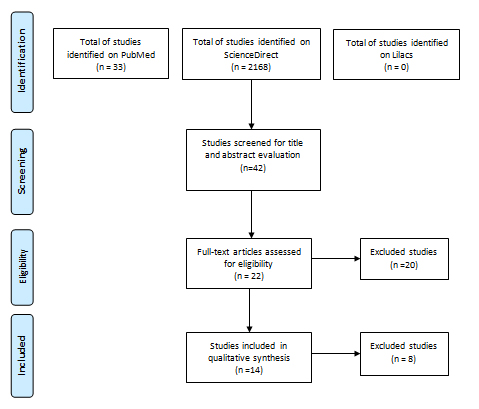
A total of 2,201 articles were found. Five independent researchers evaluated the titles and abstracts of the articles, 42 studies were pre-selected for presenting potential eligibility to the topic. In the end, 14 articles were considered relevant for the systematic review.
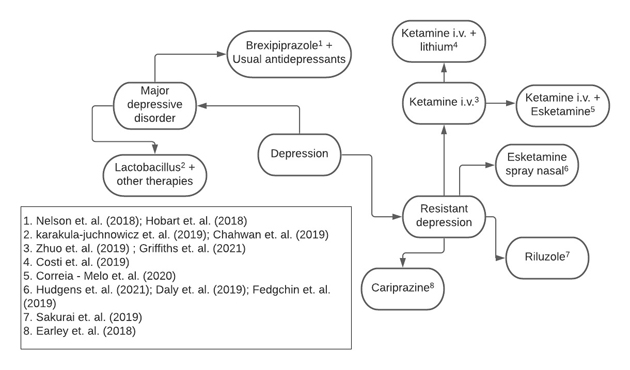
The articles included studies by Fabbri et. al. (2021) - a study about potential new treatments for TRD; Griffiths et.al. (2021) - a study about the evaluation of ketamine's antidepressant and adverse effects on TRD; Hudgens et. al. (2021) - a study about the response of esketamine nasal spray in patients with TRD; Melo et. al. (2020) - a study about the possible inferiority of esketamine compared to ketamine; Chahwan et. al (2019) - a study about the probiotic's ability to act on depressive symptoms; Daly et.al. (2019) - a study about antidepressant use associated with esketamine nasal spray in patients with TRD; Costi et. al. (2019) - a study about lithium preventing severe depression after cessation of ketamine use; Fedgchin et. al. (2019) - a study to evaluate esketamine nasal spray in TRD patients; Karakula-Juchnowicz et. al. (2019) - a study about probiotic supplementation in patients with MDD; Sakurai et. al. (2019) - a study about prolonged use of riluzole in patients with TRD; Zhuo et. al. (2019) - a study about ketamine's ability to reverse depression and synaptic loss; Earley et. al. (2018) - a study about the effect of cariprazine associated with ADT in patients with MDD; Hobart et. al. (2018) - a study about the efficacy and safety of brexpiprazole in MDD; Nelson et. al. (2018) - a study about the effects of brexpriprazole associated with ADT in patients with MDD. The main characteristics of the selected studies were summarized in Table 1.
Table 1. Summary of authors, journals, locations, objectives, methodologies, results and main findings of the studies included for analysis in the systematic review based on the PRISMA method.
3.1. Evaluation of the methodological quality of the included studies
The most common methodological deficiencies in all studies arose from insufficient reporting: samples, scenarios and recruitment procedures were often not fully described.
3.2. Mental health interventions
Ten studies reported the urgent need to implement interventions related to new pharmacological therapeutic approaches for depressive disorders. Four studies demonstrate the need for interventions for organizational adjustments through new pharmacological therapeutic approaches for depressive disorders in the last four years.
MDD is a complex, disabling mental illness and a highly prevalent health problem worldwide. Therefore, brexpripazole and the probiotic Winclove's Ecologic® Barrier were seen as new viable treatments in addition to the usual therapy. Regarding TRD, it is a pathology classified from the therapeutic failure of at least two or three trials with antidepressants of adequate dose and duration. Thus, the new medications investigated for the treatment of TRD were nasal and IV esketamine and, IV ketamine, riluzole, and cariprazine, drugs with the potential to aid in TRD (Figure 2).
4. DISCUSSION
Patients diagnosed with MDD may not respond or poorly respond to the drugs used for this pathology (Griffiths et. al., 2021). When this happens, other therapeutic strategies should be considered as a change from the current antidepressant or association with a second pharmacological agent (Nelson et. al., 2018). Given this, there was a need for studies with new agents, or the reuse of agents used therapeutically in other clinical conditions, for depression.
4.1 Major Depressive Disorder (MDD)
Brexpiprazole is a drug in the class of atypical antipsychotics that acts as a modulator of serotonin-dopamine activity, and that showed efficacy in the adjunctive treatment of MDD when in combination with antidepressants of different classes, in those patients who responded inadequately to ADT (Hobart et. al., 2018). Brexpiprazole 1 mg/day, when associated with ADT for six weeks, showed a significant improvement in the Montgomery-Asberg Depression Rating Scale (MADRS) score, positively impacting the symptoms of apparent and reported sadness, internal tension, tiredness, inability to feel, and thoughts pessimists (Nelson et. al., 2018).
The intestinal microbiota and its role in the gut-brain axis have been widely studied for several psychiatric pathologies including MDD. The main factors that generate the appearance of this depressive disorder are related to the inflammatory-immune imbalance, and the modulation of this axis can be a promising therapeutic target for mental health. (Karakula-Juchowicz et. al., 2019). The use for eight weeks of the probiotic Winclove's Ecologic® Barrier, which is composed of nine bacterial strains: Bifidobacterium bifidum W23, Bifidobacterium lactis W51, Bifidobacterium lactis W52, L. acidophilus W37, Lactobacillus brevis W63, Lactobacillus casei W56, Lactobacillus salivarius W24, Lactococcus lactis W19 e Lactococcus lactis W58, was used as a treatment in mild to moderate MDD. This medication in monotherapy was not effective in reducing depressive symptoms. However, when associated with Cognitive Behavioral Therapy, it can make changes in the patterns of cognitive reactivity concerning the sad mood associated with depression. Therefore, it can be effective when used to complement and potentiate the symptoms effects of other treatments for depressive disorders. (Chahwan et. al., 2019).
4.2 Treatment Resistant Depression (TRD)
Ketamine is a dissociative anesthetic that, in recent studies, is promising in the treatment of TRD. This drug represents new hope for those who, until then, have not responded with the combination of two or more antidepressants (Zhuo et. al., 2019). Esketamine, the S enantiomer of ketamine, is an antagonist of the N-methyl-d-aspartate (NMDA) receptor, and was recently approved by the Food and Drug Administration (FDA), in the form of a nasal spray, as pharmacological therapy in TRD (Daly et. al., 2019; Hudgens et. al., 2020; Griffiths et. al., 2021).
The choice of the ketamine S enantiomer concerning R is because it has greater affinity for the NMDA receptor than this one and, therefore, is considered more potent. The increase in the levels of glutamate in the synaptic cleft, provided by ketamine, consequently increases the number of synapses, which are reduced in depression. Such action of ketamine through the glutamatergic pathway is already pointed out as well established by the most recent studies, representing one of the mechanisms responsible for the quick antidepressant responses of the drug, since such action reverses the chronic loss of synapses that occurs in the disease (Fabbri et. al ., 2020; Zhuo et. al., 2019).
One study noted the change in MADRS scores in patients who had TRD. These patients started using a nasal spray containing 28mg of esketamine, administered twice a week, associated with the daily use of a new oral antidepressant that had not yet been used and that was one of the options of choice: duloxetine, sertraline, escitalopram or venlafaxine from prolonged action, and were compared with the placebo group that received placebo nasal spray and oral antidepressant. There was a significant improvement with the use of esketamine nasal spray, defined as a reduction higher than or equal to 50%, or a reduction of 10 points, in MARDS, influencing both the severity of symptoms and their frequency (Daly et. al., 2019; Hudgens et. al., 2020).
The adverse effects of pharmacological therapy were not significant and some patients experienced vertigo, headache, and dissociation as the most common changes (Daly et. al., 2019; Fedgchin et. al., 2019). Although pharmacological therapy with esketamine nasal spray has shown beneficial results in the short term, its long-term effects are still unclear, and it is necessary to evaluate the efficacy of the drug in this context (Daly et. al., 2019). Esketamine was also studied in its IV form to compare its effectiveness with that of IV ketamine. In this study, the rate of remission of depressive symptoms in 24 hours of IV esketamine 0.25 mg/kg was 24.1% and that of IV ketamine 0.5 mg/kg 29.1%, thus not lower than ketamine and presenting both efficacy, tolerability, and safety (Correia-Melo et. al., 2020).
A phenomenological analysis was done with 13 patients with TRD who used IV ketamine, in an individual dose calculated according to height and weight. The doses administered ranged from 25 mg/40 ml to 75 mg/40 ml, all infused over 40 minutes, during three to 112 sessions. Interviews were conducted with these patients to assess the effects of the medication. For many of them, feelings such as “happy and laughing”, “more willing to do activities”, “greater socialization and communication with others”, “feeling normal”, “with positive thoughts” and “reduced suicidal ideation” were reported (Griffiths et. al., 2021).
Ketamine emerges as an effective therapy and able to work in different neurobiological pathways to provide its antidepressant actions. The drug can work in the circadian cycle, prolonging REM sleep time and increasing levels of brain-derived neurotrophic factor (BDNF) responsible for regulating slow-wave sleep. Thus, being able to redefine the changes in circadian rhythm caused by depression and improve synaptic plasticity is also affected by the disease. (Fabbri et. Al., 2020; Zhuo et. Al., 2019)
The action of ketamine in modulating inflammation and the immune system as an inhibitor of glycogen synthase kinase 3 beta (GSK3B) that modulates the activity of proteins responsible for mood regulation, also seems to be responsible for the rapid antidepressant effects of ketamine (Fabbri et al., 2020). Lithium, which is also an inhibitor of (GSK3), was the target of a randomized double-blind study that aimed to assess whether this drug was able to prolong the antidepressant effects of ketamine. The participants received IV ketamine and, then, associated with lithium 600 to 1200 mg/day orally or with placebo for a week, and finally maintained with only lithium or placebo. However, no significant difference was seen between the groups. The addition of lithium did not, therefore, appear to have led to a reduction in MADRS scores compared to treatment with ketamine alone. (Costi et. al., 2019).
In addition to ketamines, other drugs are targets for studies for patients with TRD and patients with inadequate response to ADT. Studies conducted with riluzole and cariprazine in these patients used MADRS to assess primary efficacy. The mechanism of action of riluzole is believed to be the inhibition of presynaptic release of glutamate through the inactivation of voltage-gated sodium channels in glutamatergic nerve terminals, it is a drug used to treat Amyotrophic Lateral Sclerosis (ALS) and has been evaluated as long-term therapy in patients with TRD. Riluzole showed positive results, as it can promote a reduction higher than or equal to 50% in MADRS, despite not showing great differences concerning the placebo in terms of efficacy for the treatment of patients with TRD. The study realized with cariprazine, an atypical antipsychotic whose main mechanism of action is the partial agonist of dopamine D2 and D3 receptors, in patients with inadequate response to the usual treatment with antidepressants, did not show any significant changes in MADRS or a considerable decrease in depressive symptoms when faced with placebo. Consequently, further studies should be carried out to better understand these drugs in the treatment of depression. (Sakurai, et. al., 2019; Earley, et. al., 2018)
5.CONCLUSION
Knowing that 20-30% of patients with MDD develop TRD (Fabbri et. al., 2020), the new therapies under study appear to improve the approach of these patients and prevent disease progression.
According to this systematic review, the use of brexpiprazole associated with antidepressants of different classes, as well as the use of Winclove's Ecologic® Barrier-type lactobacilli for eight weeks, showed short-term efficacy in the treatment of MDD. On the other hand, pharmacological options, such as ketamine, esketamine, riluzole, and cariprazine, appear as new possibilities for those who until then have not obtained remission of the depressive disorder with the use of more than two antidepressants.
As assessed by Griffiths et. al. (2021) and Fabbri et. al. (2020), although the new pharmacological options addressed in our study have shown efficacy in the treatment of depressive disorders, we realize that there are still gaps in both the knowledge of the action of drugs at the neurobiological level associated with the neurophysiology of the disease and the action of these drugs over the long term.
REFERENCES
Chahwan, B., Kwan, S., Isik, A., Hemert, S.V., Burke, C., Roberts, L., 2019. Gut feelings: A randomised, triple-blind, placebo-controlled trial of probiotics for depressive symptoms. Journal of Affective Disordens, 253, 317-326. https://doi.org/10.1016/j.jad.2019.04.097 .
Correia-Melo, F.S, Leal, G.C., Vieira, F., Nunes, A.P.J., Mello, R.P., Magnavita, G., et. al., 2020. Efficacy and safety of adjunctive therapy using esketamine or racemic ketamine for adult treatment-resistant depression: A randomized, double-blind, non-inferiority study. Journal of Affective Disorders, 264, 527-534. https://doi.org/10.1016/j.jad.2019.11.086 .
Costi, S., Soleimani, L., Glasgow, A., Brallier, J., Spivack, J., Schwartz, J., et. al., 2019. Lithium continuation therapy following ketamine in patients with treatment resistant unipolar depression: a randomized controlled trial. Neuropsychopharmacology, 44, 1812-1819. http://doi.org/10.1038/s41386-019-0365-0 .
Daly, E.J., Trivedi, M.H., Janik, A., Li, H., Zhang, Y., Li, X., et. al., 2019. Efficacy of Esketamine Nasal Spray Plus Oral Antidepressant Treatment for Relapse Prevention in Patients With Treatment-Resistant Depression: A Randomized Clinical Trial. JAMA Psychiatry, 76, 893-903. https://doi.org/10.1001/jamapsychiatry.2019.1189 .
Earley, W.R., Gou, H., Németh, G., Harsányi, J., Thase, M.E., 2018. Cariprazine Augmentation to Antidepressant Therapy in Major Depressive Disorder: Results of a Randomized, Double- Blind, Placebo-Controlled Trial. Psychopharmacology Bulletin, 48, 62-80. PMCID: PMC6294423. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6294423/pdf/PB-48-4-62.pdf .
Fabbri, C., Kasper, S., Zohar, J., Souery., D., Montgomery, S., Albani, D., et. al., 2021. Drug repositioning for treatment-resistant depression: Hypotheses from a pharmacogenomic study. Progress in Neuropsychopharmacology & Biological Psychiatry, 104, 110050. https://doi.org/10.1016/j.pnpbp.2020.110050 .
Fedgchin, M., Trivedi, M., Daly, E.J., Melkote, R., Lane, R., Lim, P., et. al., 2019. Efficacy and Safety of Fixed-Dose Esketamine Nasal Spray Combined With a New Oral Antidepressant in Treatment- Resistant Depression: Results of a Randomized, Double-Blind, Active-Controlled Study (TRANSFORM-1). International Journal of Neuropsychopharmacology, 22, 616-630. https://doi.org/10.1093/ijnp/pyz039 .
Griffiths, C., Walker, K., Reid, I., Silva, K.M., O´Neill-Kerr, A., 2021. A qualitative study of patients ´ experience of ketamine treatment for depression: The ´Ketamine and me´project. Journal of Affective Disordens Reports, 4, 100079. https://doi.org/10.1016/j.jadr.2021.100079 .
Karakula-Juchnowicz, H., Rog, J., Juchnowicz, D., Loniewski, I., Żydecka, K.S., Krukow, P., et. al., 2019. The study evaluating the effect of probiotic supplementation on the mental status, inflammation, and intestinal barrier in major depressive disorder patients using gluten-free or gluten-containing diet (SANGUT study): a 12-week, randomized, double-blind, and placebo-controlled clinical study protocol. Nutrition Journal,18, 50. https://doi.org/10.1186/s12937-019-0475-x .
Hobart, M., Skuban, A., Zhang, P., Augustine. C., Brewer, C., Hefting, N., et. al., 2018. A Randomized, Placebo-Controlled Study of the Efficacy and Safety of Fixed-Dose Brexpiprazole 2 mg/d as Adjunctive Treatment of Adults With Major Depressive Disorder. Journal of Clinical Psychiatry, 79. https://doi.org/10.4088/JCP.17m12058 .
Hudgens, S., Floden, L., Blackowicz, M., Jamieson, C., Popova, V., Fedgchin, M., et. al., 2021. Meaningful Change in Depression Symptoms Assessed with the Patient Health Questionnaire (PHQ-9) and Montgomery- Asberg Depression Rating Scale (MADRS) Among Patients with Treatment Resistant Depression in Two, Randomized, Double-blind, Active-controlled Trials of Esketamine Nasal Spray Combined With a New Oral Antidepressant. Journal of Affective Disorders, 281, 767-775. https://doi.org/10.1016/j.jad.2020.11.066 .
Nelson, J.C., Weiller, E., Zhang, P., Weiss, C., Hobart, M., 2018. Efficacy of adjunctive brexpiprazole on the core symptoms of major depressive disorder: A post hoc analysis of two pooled clinical studies. Journal of Affective Disorders, 227, 103-108. https://doi.org/10.1016/j.jad.2017.09.054 .
Sakurai, H., Dording, C., Yeung, A., Foster, S., Jain, F., Chang, T., et. al., 2019. Longer-term open-label study of adjunctive riluzole in treatment-resistant depression. Journal of Affective Disorders, 258, 102-108. https://doi.org/10.1016/j.jad.2019.06.065 .
Zhuo, C., Tian, H., Li, G., Chen, M., Jiang, D., Lin, X., et. al., 2019. Effects of ketamine on circadian rhythm and synaptic homeostasis in patients with treatment-resistant depression: A protocol for mechanistic studies of its rapid and sustained antidepressant in humans. Brain and Behavior, 9. https://doi.org/10.1002/brb3.1423 .








